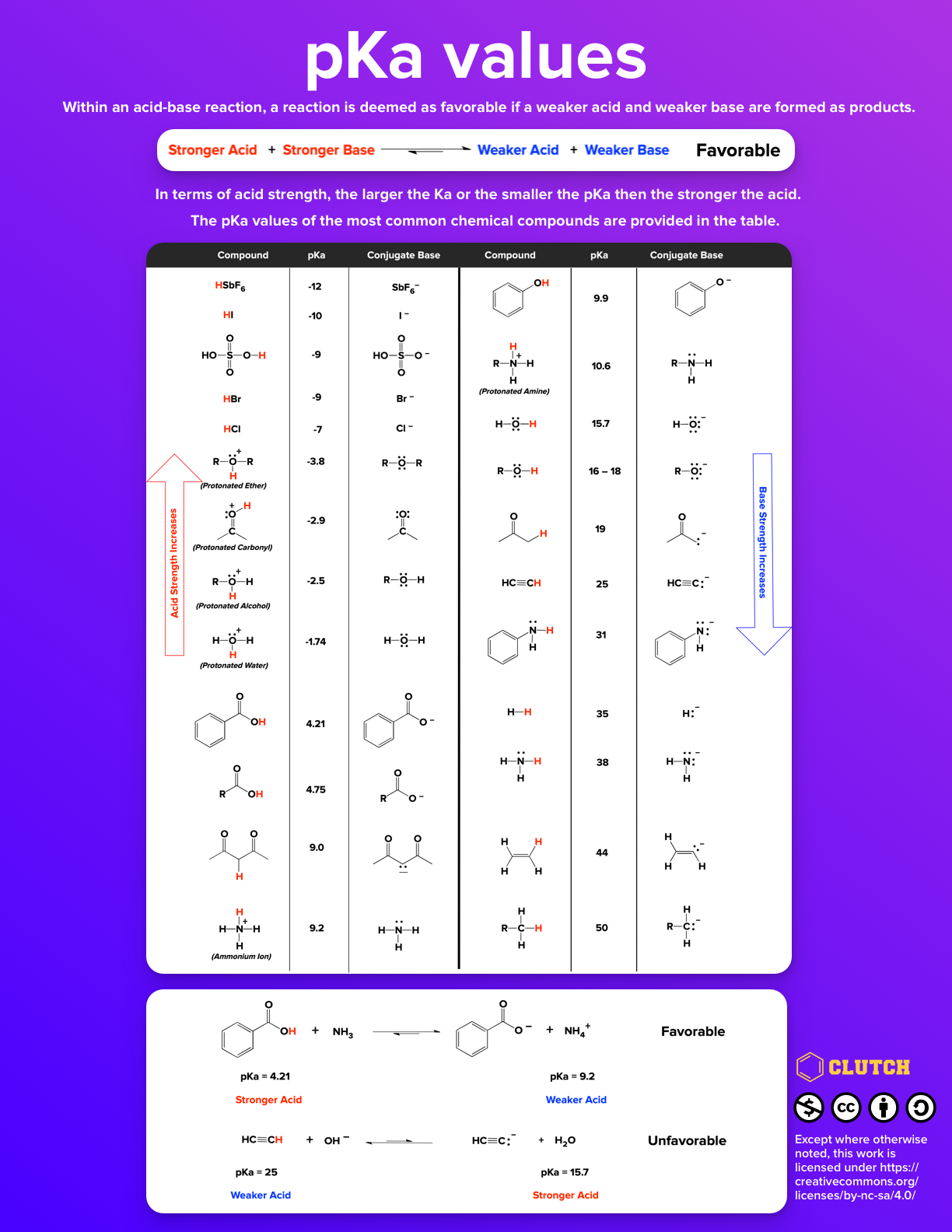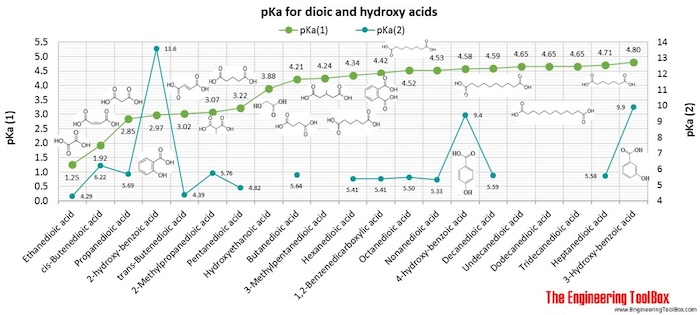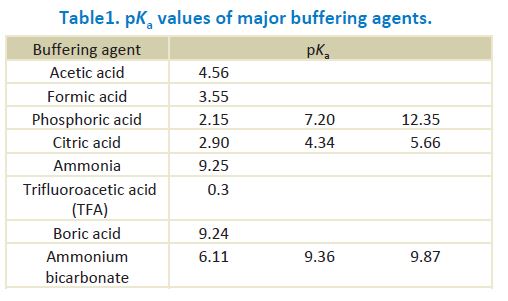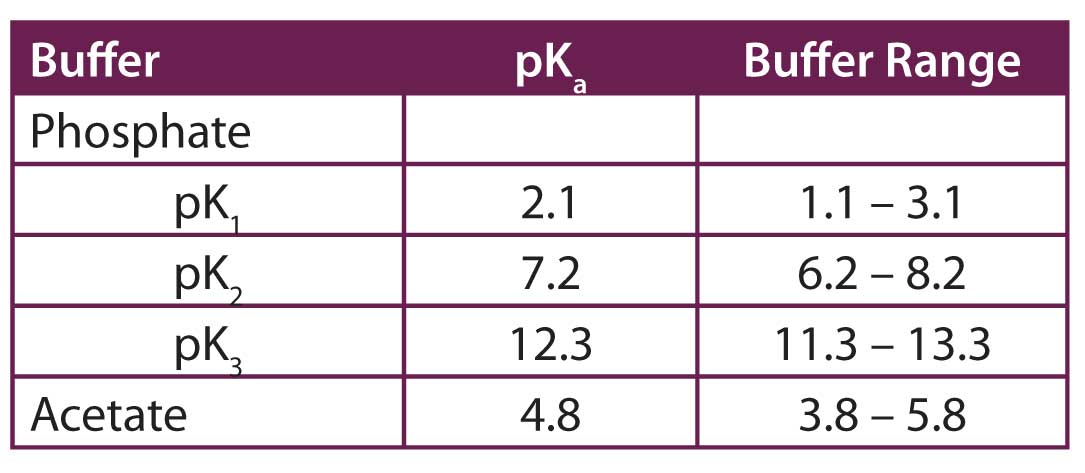
Breaking old habits: Moving away from commonly used buffers in pharmaceuticals - European Pharmaceutical Review

Chemical composition of some buffer solutions covering the pH range of... | Download Scientific Diagram

Solution pKa values for a range of polymers at various polymer and salt... | Download Scientific Diagram

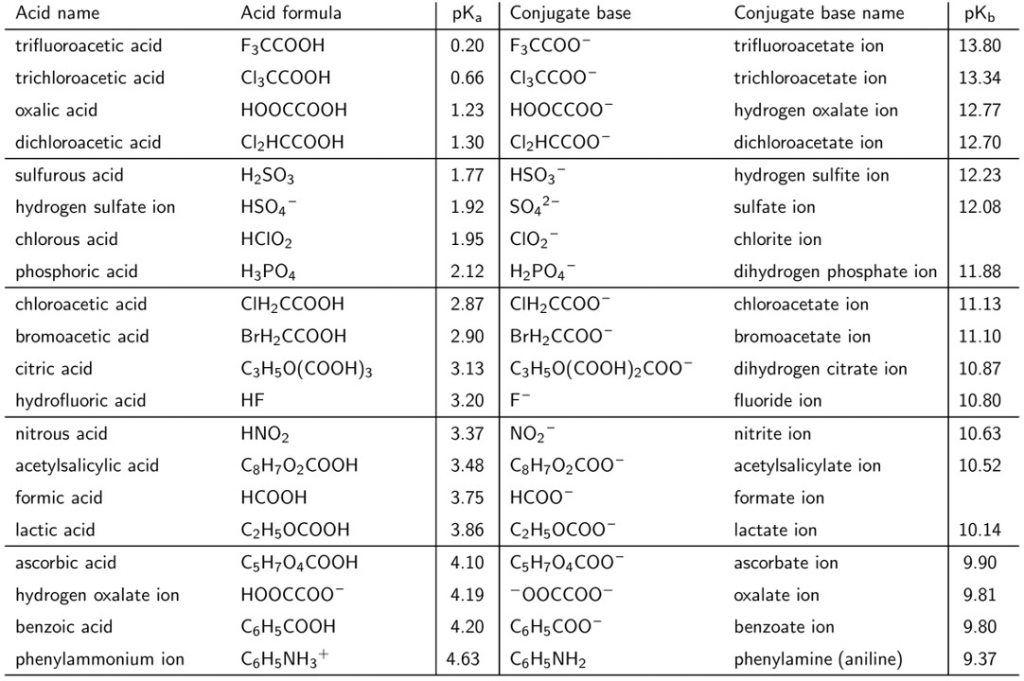
Table 4 from Measurement of dissociation constants (pKa values) of organic compounds by multiplexed capillary electrophoresis using aqueous and cosolvent buffers. | Semantic Scholar

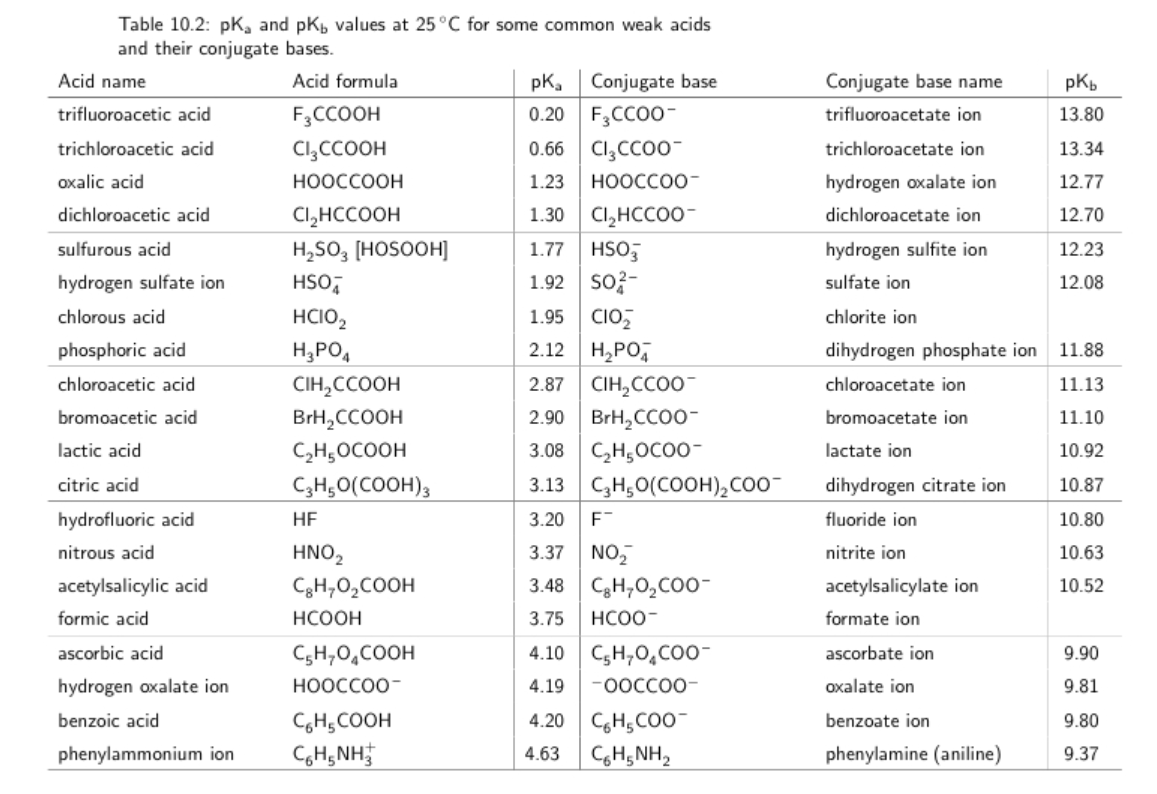
SOLVED: You are trying to create a buffer with pH = 7.7. a.) Which acid from the table below should you use? b.) What else do you need to add to create

equilibrium - Why do buffers need to be composed of equal amounts of the acid and salt? - Chemistry Stack Exchange

Breaking old habits: Moving away from commonly used buffers in pharmaceuticals - European Pharmaceutical Review

Using the weak acid pKa data shown in the following table, how would you prepare 1.0L of a 2.0M buffer with - brainly.com
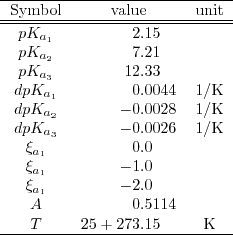
![PDF] Buffer solutions in drug formulation and processing: How pKa values depend on temperature, pressure and ionic strength | Semantic Scholar PDF] Buffer solutions in drug formulation and processing: How pKa values depend on temperature, pressure and ionic strength | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/879d3eae13956041f4cc1b8ee223109275087b7e/3-Table1-1.png)
PDF] Buffer solutions in drug formulation and processing: How pKa values depend on temperature, pressure and ionic strength | Semantic Scholar

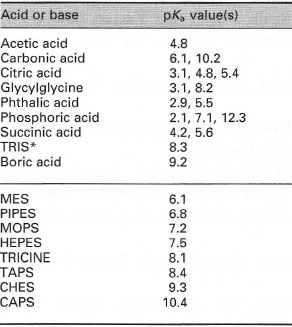
SOLVED: 11. Buffer preparation Given the KalpKa of the following compounds: TABLE 8.3 Some K; and pK; Values for Weak Acids, in Decreasing Order of Strength Name Formula K pK. Phosphorie acid